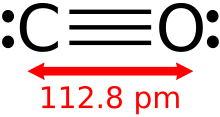
A monoxide is any oxide containing only one atom of oxygen. A well known monoxide is carbon monoxide; see carbon monoxide poisoning.
The prefix mono (Greek for "one") is used in chemical nomenclature.[1] In proper nomenclature, the prefix is not always used in compounds with one oxygen atom.[2] Generally, when the oxygen is bonded to a nonmetal, the prefix mono is used. However when the oxygen atom bonds to a metal, the prefix is dropped. For instance, in the compound K2O, potassium (K) is a metal and therefore its proper name is potassium oxide, rather than potassium monoxide.
Among monoxides, carbon monoxide and hydrogen monoxide (water) are both neutral, germanium(II) oxide is distinctly acidic, and both tin(II) oxide and lead(II) oxide are amphoteric.
YouTube Encyclopedic
-
1/3Views:5 9211 540 28140 903
-
SYMPTOMS AND CAUSES OF CARBON MONOXIDE POISONING - PLUS HOW TO PREVENT IT
-
Destroying A Dangerous Yellowjacket Nest Using Carbon Monoxide.
-
How Carbon Monoxide Poisoning works
Transcription
References
- ^ Foundations of College Chemistry, 13th Edition. Wiley. 2010. p. 110. ISBN 978-0470460610.
- ^ Nomenclature of Inorganic Chemistry, IUPAC Recommendations 2005. 2005. pp. 69, 70. ISBN 0-85404-438-8.
